5 results
Esketamine nasal spray shows higher remission and response rates over 32 weeks of treatment compared with quetiapine extended-release in patients with treatment resistant depression: Results from ESCAPE-TRD, a randomised, phase IIIb clinical trial
- A. Reif, A. E. Anıl Yağcıoğlu, A. Luts, T. Messer, R. Nielsen, J. Buyze, T. Ito, Y. Kambarov, S. Mulhern Haughey, B. Rive, I. Usankova, C. von Holt, Y. Godinov
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S90-S91
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Treatment resistant depression (TRD) is estimated to affect 10–30% of patients with major depressive disorder (Al‑Harbi et al. Patient Prefer Adherence 2012; 6 369–88). Esketamine nasal spray (NS), in combination with a selective serotonin reuptake inhibitor (SSRI) or serotonin norepinephrine reuptake inhibitor (SNRI), increases remission and response rates in patients with TRD compared with placebo plus SSRI/SNRI (Popova et al. Am J Psychiatry 2019; 176 428–38). ESCAPE-TRD (NCT04338321) is the first randomised clinical trial to compare esketamine NS to quetiapine extended-release (XR), an antipsychotic augmentation therapy for patients with TRD.
ObjectivesTo explore the efficacy and safety of esketamine NS compared with quetiapine XR in TRD over 32 weeks (wks).
MethodsIn the ESCAPE-TRD phase IIIb open-label, rater-blinded trial, patients were randomised 1:1 to esketamine NS (56/84 mg; twice per wk, weekly or every 2 wks) or quetiapine XR (150–300 mg daily) both in combination with an ongoing SSRI/SNRI. Remission (Montgomery-Åsberg Depression Rating Scale [MADRS] total score of ≤10) and response (≥50% improvement in MADRS total score from baseline or MADRS≤10) rates were analysed over time using last observation carried forward. MADRS change from baseline was analysed using Mixed Models for Repeated Measures (MMRM). The most common adverse events (AEs) leading to discontinuation are reported for patients who received ≥1 dose of study medication.
ResultsAt baseline, 336 patients were randomised to esketamine NS and 340 to quetiapine XR. A significantly higher percentage of patients in the esketamine NS group achieved remission (at each visit from Wk6 [p=0.008] onward) and response (at each visit from Day 15 [p<0.001] onward) versus patients treated with quetiapine XR. Esketamine NS significantly improved MADRS score compared to quetiapine XR at each visit from Day 8 onwards, with an average difference over time in the least squares means total MADRS score change from baseline of -2.4 (Figure). The most common AEs leading to treatment discontinuation for esketamine NS were dizziness (n=2, 0.6%), dissociation (n=2, 0.6%) and vomiting (n=2, 0.6%), and for quetiapine XR were sedation (n=7, 2.1%), weight increased (n=6, 1.8%) and somnolence (n=5, 1.5%).
Image:
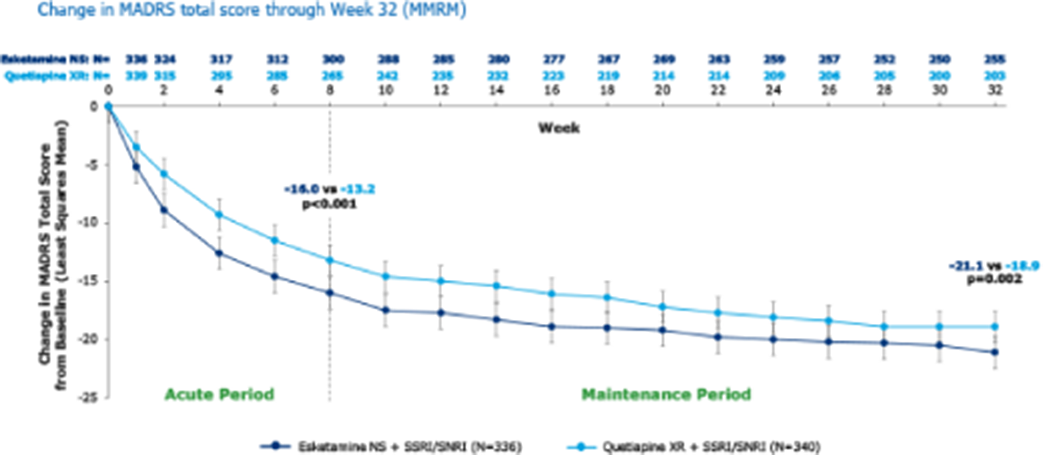 Conclusions
ConclusionsEsketamine NS increased the percentage of patients achieving response and remission and improved MADRS total score over time compared with quetiapine XR. Rates of discontinuation arising from the most common AEs were generally lower with esketamine NS than quetiapine XR.
AcknowledgementsWe thank participating patients and all who assisted with the study. This study was funded by Janssen; medical writing support was provided by Carolyn Walsh, PhD, Costello Medical, UK.
Disclosure of InterestA. Reif Grant / Research support from: Medice, Consultant of: National Care Guidelines (NVL, S3) on major depression, bipolar disorder, ADHD and suicidal behaviour (aided in developing guidelines); board member of DGBS, DGPPN, ECNP and German Depression Foundation, Speakers bureau of: (and participated in advisory boards over the last 3 years) for Cyclerion, Janssen, Medice, SAGE/Biogen and Shire/Takeda; received speaker’s honoraria from Das Fortbildungskolleg; , A. E. Anıl Yağcıoğlu Grant / Research support from: Participated as an investigator for Janssen, Speakers bureau of: (and participated in advisory boards over the last 3 years) for Janssen and Abdi İbrahim Otsuka, A. Luts Speakers bureau of: (or participated in advisory boards for or participated as an investigator) for Janssen-Cilag, Asarina Pharma, Bristol Meyer Squibb, Dr August Wolff GmbH & Co, Eli Lilly, Lundbeck, Pfizer, Allergan, Sunovion and Regeneron., T. Messer Consultant of: National Care Guidelines (NVL, S3) on major depression (aided in developing guidelines), Speakers bureau of: (and participated in advisory boards) for Janssen-Cilag and Otsuka/Lundbeck, R. Nielsen Consultant of: Board member of DSAL and IGSLi, Speakers bureau of: (or participated in advisory boards, received research funds or participated as investigator over the last 3 years) for Boehringer Ingelheim, Compass Pharmaceuticals, Janssen-Cilag, Lundbeck, Otsuka, Sage and Teva Pharmaceuticals, J. Buyze Employee of: Janssen, T. Ito Employee of: Janssen, Y. Kambarov Employee of: Janssen, S. Mulhern Haughey Employee of: Janssen, B. Rive Employee of: Janssen, I. Usankova Employee of: Janssen, C. von Holt Employee of: Janssen, Y. Godinov Employee of: Janssen
Esketamine Nasal Spray Improves Rate and Time to Remission Versus Quetiapine Extended Release in Subgroups of Patients With Treatment Resistant Depression and Two or Three Plus Prior Treatment Failures: Results From ESCAPE-TRD, a Randomised Phase IIIb Trial
- A.H. Young, W.J. Cubała, R.E. Nielsen, A. Popova, T. Ito, S. Mulhern-Haughey, N. Pirotte, B. Rive, P. Thilakarathne, I. Usankova, Y. Godinov
-
- Journal:
- BJPsych Open / Volume 9 / Issue S1 / July 2023
- Published online by Cambridge University Press:
- 07 July 2023, p. S75
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Aims
For patients with depression, the likelihood of remission decreases with each subsequent treatment failure. Per European Medicines Agency guidance, treatment resistant depression (TRD) is defined as nonresponse to ≥2 consecutive treatments at adequate dosage and duration in the current depressive episode. In ESCAPE-TRD (NCT04338321), esketamine nasal spray (NS) increased the probability of achieving remission and remaining relapsefree, compared with quetiapine extended release (QXR) in patients with TRD. Here, we report the efficacy of esketamine NS vs QXR in patient subgroups with 2 or ≥3 consecutive prior treatment failures (PTFs).
MethodsESCAPETRD was a phase IIIb trial comparing the efficacy of esketamine NS with QXR in patients with TRD. Patients (N = 676) were randomised 1:1 to esketamine NS (n = 336; 56/84 mg; twice weekly, weekly, or every 2 weeks [wks]) or QXR (n = 340; 150–300 mg daily, both in combination with an ongoing selective serotonin reuptake inhibitor/serotonin norepinephrine reuptake inhibitor. Randomisation was stratified by age (18-64 years; 65–74 years) and PTFs (2; ≥3).
The primary endpoint of remission (Montgomery-Åsberg Depression Rating Scale total score ≤10) at Wk8 and the secondary endpoint of remaining relapse-free through Wk32 after remission at Wk8, were analysed in PTF patient subgroups and compared between study arms, with treatment discontinuation considered as a negative outcome. The effect on time to remission was assessed using hazard ratios (HR) from a Cox regression model.
ResultsOf the randomised patients, 415 (61.4%; esketamine NS: 204, QXR: 211) had experienced 2 PTFs and 261 (38.6%; esketamine NS: 132, QXR: 129) had experienced ≥3.
Of patients with 2 PTFs, 54/204 (26.5%) esketamine NS-treated patients and 46/211 (21.8%) Q-XR-treated patients achieved remission at Wk8 (p = 0.267). Of patients with ≥3 PTFs, 37/132 (28.0%) and 14/129 (10.9%) patients achieved remission at Wk8 in esketamine NS and Q-XR arms, respectively (p < 0.001). Of patients with 2 and ≥3 PTFs, 49/204 (24.0%) and 24/132 (18.2%) of esketamine NS-treated patients and 38/211 (18.0%) and 10/129 (7.8%) of Q-XR-treated patients achieved remission at Wk8 without relapse to Wk32 (p = 0.133 and p = 0.013), respectively.
Esketamine NS significantly improved time to remission, with a greater effect in the ≥3 PTF subgroup (2 PTFs: HR = 1.547 [95% confidence interval (CI) 1.210–1.976]; p < 0.001 vs ≥3 PTFs: HR = 2.066 [95% CI 1.469–2.907]; p < 0.001).
ConclusionEsketamine NS demonstrated a significantly superior remission rate versus QXR at Wk8 in patients with ≥3 PTFs, and significantly shorter time to remission versus Q-XR in both subgroups.
Gastrostomy tube placement in congenital cardiac surgery: a multi-institutional database study
- Brena S. Haughey, Peter Dean, Michael C. Spaeder, Clyde J. Smith, Mark Conaway, Shelby C. White
-
- Journal:
- Cardiology in the Young / Volume 33 / Issue 9 / September 2023
- Published online by Cambridge University Press:
- 03 October 2022, pp. 1672-1677
-
- Article
- Export citation
-
Introduction:
Neonates and infants who undergo congenital cardiac surgery frequently have difficulty with feeding. The factors that predispose these patients to require a gastrostomy tube have not been well defined. We aimed to report the incidence and describe hospital outcomes and characteristics in neonates and infants undergoing congenital cardiac surgery who required gastrostomy tube placement.
Materials and method:A retrospective review was performed on patients undergoing congenital cardiac surgery between October 2015 and December 2020. Patients were identified by International Classification of Diseases 10th Revision codes, utilising the performance improvement database Vizient® Clinical Data Base, and stratified by age at admission: neonates (<1 month) and infants (1–12 months). Outcomes were compared and comparative analysis performed between admissions with and without gastrostomy tube placement.
Results:There were 11,793 admissions, 3519 (29.8%) neonates and 8274 (70.2%) infants. We found an increased incidence of gastrostomy tube placement in neonates as compared to infants following congenital cardiac surgery (23.1% versus 6%, p = <0.001). Outcomes in neonates and infants were similar with increased length of stay and cost in those requiring a gastrostomy tube. Gastrostomy tube placement was noted to be more likely in neonates and infants with upper airway anomalies, congenital abnormalities, hospital infections, and genetic abnormalities.
Discussion:Age at hospitalisation for congenital cardiac surgery is a definable risk factor for gastrostomy tube requirement. Additional factors contribute to gastrostomy tube placement and should be used when counselling families regarding the potential requirement of a gastrostomy tube.
Clinical consensus regarding the importance of rapid reduction in depressive symptoms in major depressive disorder with acute suicidal ideation or behavior (MDSI)
- S. Borentain, A. Nash, E. Daly, K. Joshi, M. O’Hara, Q. Zhang, M. Mathews, S. Haughey, S. Richards, J. Anjo, D. Zante, R. Perry
-
- Journal:
- European Psychiatry / Volume 65 / Issue S1 / June 2022
- Published online by Cambridge University Press:
- 01 September 2022, p. S558
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Patients with major depressive disorder (MDD) with acute suicidal ideation or behavior (MDSI) require immediate intervention. Though oral antidepressants can be effective at reducing depressive symptoms, they can take 4–6 weeks to reach full effect.
ObjectivesThis study aimed to identify unmet needs in the treatment of patients with MDSI, specifically exploring the potential clinical benefits of rapid reduction of depressive symptoms.
MethodsA Delphi panel consisting of practicing psychiatrists (n=12) from the US, Canada and EU was conducted between December 2020–June 2021. Panelists were screened to ensure they had sufficient experience with managing patients with MDD and MDSI. Panelists completed two survey rounds, and a virtual consensus meeting.
ResultsThis research confirmed current unmet needs in the treatment of patients with MDSI.
Hopelessness, functional impairment, worsening of MDD symptoms, recurrent hospitalization and higher risk of suicide attempt were considered as key consequences of the slow onset of action of oral antidepressants.
Treatment with rapid acting antidepressant was anticipated by panelists to provide short-term benefit such as rapid reduction of core MDD symptoms which may contribute to shorter hospital stays and improved patient engagement/compliance, allowing for earlier interventions and improved patient outcomes. For long-term benefits, panelists agreed that improved daily functioning and increased trust/confidence in treatment options, constitute key benefits of rapid-acting treatments
ConclusionsThere is need for rapid-acting treatments which may help address key unmet needs and provide clinically meaningful benefits driven by the rapid relief of depressive symptoms particularly in patients with MDSI.
DisclosureSB, ED, KJ, MO’H, QZ, MM, MH, SR, JA and DZ are employees of Janssen and hold stock in Johnson & Johnson Inc. AN is currently employed by Neurocrine Biosciences Inc. RP is an employee of Adelphi Values PROVE hired by Janssen.
Contributors
-
- By Robert S. Anderson, (Mary) Colleen Bhalla, Michelle Blanda, Christopher Carpenter, Chris Chauhan, Paul L. DeSandre, Maura Dickinson, Jonathan A. Edlow, Dany Elsayegh, Kara Iskyan Geren, Peter J. Gruber, Jin H. Han, Marianne Haughey, Teresita M. Hogan, Ula Hwang, Lindsay Jin, Michael P. Jones, Joseph H. Kahn, Keli M. Kwok, Denise Law, Megan M. Leo, Stephen Y. Liang, Judith A. Linden, Brendan G. Magauran Jr, Joseph P. Martinez, Amal Mattu, Karen M. May, Aileen McCabe, Kerry K. McCabe, Jolion McGreevy, Ron Medzon, Ravi K. Murthy, Aneesh T. Narang, Lauren M. Nentwich, David E. Newman-Toker, Jonathan S. Olshaker, Joseph R. Pare, Thomas Perera, Joanna Piechniczek-Buczek, Jesse M. Pines, Timothy Platts-Mills, Suzanne Michelle Rhodes, Lynne Rosenberg, Mark Rosenberg, Todd C. Rothenhaus, Kristine Samson, Arthur B. Sanders, Jeffrey I. Schneider, Rishi Sikka, Kirk A. Stiffler, Morsal R. Tahouni, Mary E. Tanski, Abel Wakai, Scott T. Wilber, Deborah R. Wong
- Edited by Joseph H. Kahn, Brendan G. Magauran, Jr, Jonathan S. Olshaker
-
- Book:
- Geriatric Emergency Medicine
- Published online:
- 05 January 2014
- Print publication:
- 16 January 2014, pp vii-x
-
- Chapter
- Export citation




